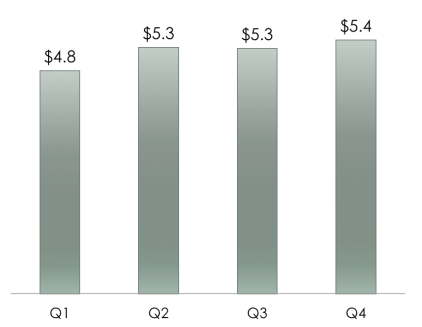
The pricing strategy for Biosimilars is still evolving. Few years ago it was assumed that Biosimilars would command low discount compared to small molecule generics. However, recent events illustrate two ends of the pricing spectrum for Biosimilars. Here we briefly discuss the two examples:
Sweden Approval of Benepali
Benepali is the biosimilar version of Amgen and Pfizer’s mega blockbuster drug Enbrel. Amgen markets Enbrel in the United States and Pfizer markets it outside the US and Canada. In 2015, Enbrel’s sales in the US and Canada were $5.4 Billion.
Today Sweden’s HTA agency approved Benepali for reimbursement at only 10% discount compared to Enbrel. This price discount is relatively low compared to typical price discounts for biosimilar ESAs and GM-CSFs.
The HTA agency’s comments for the Benepali were:
HTA believes that Benepali has the same efficacy and safety of Enbrel
Extensive analysis of Benepalis immunogenicity showed that the immune response to etanercept and its influence on the efficacy and safety comparable between Benepali and Enbrel
Norway’s Aggressive Price Negotiation for Biosimilar Remicade
Remicade is an Anti-CD 20 mAb marketed by Janssen. In 2015, Remicade’s sales were $6.6B. The biosimilar version of Remicade, called Remsima was approved in Norway after negotiating ~70% discount.